Supreme Court lifts ban on sale of Painkiller Saridon, 2 other drugs
Supreme Court lifts ban on sale of Painkiller Saridon, 2 other drugs
Mangalore Today News Network
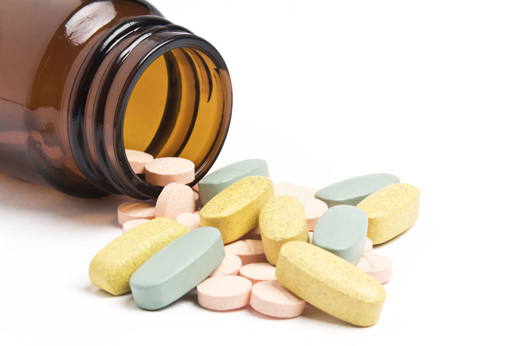
New Delhi, Sept 17, 2018 : The Supreme Court on Monday lifted ban on sale of painkiller Saridon and two other drugs.
The top court ruling comes after the September 13 order by the Ministry of Health that banned the manufacture for sale or distribution of 328 fixed-dose combination (FDC) drugs. The banned medicines included brands like Piramal Healthcare’s Saridon, Alkem Laboratories’ Taxim AZ and Macleods Pharma’s Panderm Plus cream.
A bench of Justices R F Nariman and Indu Malhotra issued notice to the Centre and sought its reply on the plea filed by some drug makers and pharma associations.
The medicines whose sale was allowed were Saridon, GlaxoSmithKline’s Piriton, Juggat Pharma’s Dart and another drug, the details of which could not be immediately known.
FDCs are two or more drugs combined in a fixed ratio into a single dosage form. The domestic pharmaceutical market size is pegged at around Rs 1.18-1.2 lakh crore, reports Business Today, and FDCs reportedly make up nearly 50% of the sales.
Public health experts and doctors in India and abroad have warned against the increasing use of antibiotic combinations as it may be contributing to antibiotic resistance.
India poses a particular concern as the market share of combination drugs versus single drugs is bigger than anywhere in the world.
An expert panel formed by the Drugs Technical Advisory Board (DTAB), in its report to the Centre, stated that there is no therapeutic justification for the ingredients contained in 328 FDCs and that these FDCs may involve risk to human beings.
DTAB said these combinations may lead to overuse. According to the technical body, there is no need to expose the patients to that many ingredients when one will do the work. The notification stated, “Hence in the larger public interest, it is necessary to prohibit the manufacture, sale or distribution of this FDC…any kind of regulation or restriction to allow for any use in patients is not justifiable.”
courtesy:News 18
Read other news
- Raymond Group’s Former Chairman Vijaypat Singhania Passes Away at 87
- Pentagon Weighs Limited Ground Operations in Iran: Report
- India denies Elon Musk was part of Trump-Modi call on West Asia war
- Iran ‘Thanks India’ with messages on missiles amid West Asia conflict
- Karnataka chief secretary reviews security ahead of IPL opener at M Chinnaswamy Stadium
- Don’t you have any shame, terrorist: Professor’s outburst against student in class
- Goa nightclub fire: Court denies bail to Luthra brothers in forgery case
- ‘Productive conversation’: White House reacts to PM Modi-Trump-Musk call on Iran war
- Bengaluru: Pregnant minor forced into marriage; case registered
- Rumours of lockdown in India completely false, no such proposal: Hardeep Singh Puri
- Excise duty on petrol, diesel slashed amid rising crude prices due to West Asia crisis
- MUDA case: High Court issues notice to CM Siddaramaiah, wife over challenge to ‘B’ report
- Cooker Bomb Blast Case: Accused Shariq Pleads Guilty, NIA Court Accepts Application
- Teen Drowns While Swimming in Belthangady Pond
- Dakshina Kannada SP Arun K selected for CM’s Medal
- Mother, Daughter Killed in Car–Truck Collision at Byndoor
- Thunder Showers Bring Relief from Heat in Coastal Districts
- Man Arrested from Manjeshwar for Communal Social Media Post
- Mangaluru University 44th convocation: Honorary doctorates conferred on Harekal Hajabba, Ravish Padumale
- Dakshina Kannada to set up stray dog shelters across all taluks
- Karnataka to replace SSLC third language marks with grading system; total marks reduced to 525
- Konkan Railway extends Surat–Mangaluru, Ahmedabad–Mangaluru special train services till May-end
- Mangaluru: Migrant worker killed as crane bucket falls during well digging in Kuthar
- Cancel licences of shops selling tobacco, drugs near schools: DK SP
- Car used in rowdy-sheeter Arif murder traced, seized in Bantwal
- Mangaluru Student Goes Missing; Ullal Police Register Case
- APD Foundation Joins WHO Civil Society Commission
- Daiva’s prediction comes true: Janardhan Reddy walks free from jail
- Skills and Competencies Take Center Stage at MSN Dialogue Series
- Court remands Maoist Lakshmi to six-day police custody
- Sandhya Shenoy honored with Society for Materials Chemistry Medal-2024
- White Cornus Apartment in Mangaluru
- City girl wins first place in state-level spell bee competition
- Alleged ‘Love Jihad’ Case in Mangaluru: Woman left home voluntarily, says police
- Girl fatally struck by reckless two-wheeler near Belman
- New residential complex for the judges inaugurated in Mangaluru
- Absconding accused nabbed after 8 years
- Truck with cylinders turns turtle in Beltangady
- Bhoota Kola artist dies of cardiac arrest
- Development of the country should be our goal: Ganesh Karnik
- CITY INFORMATION
- TRAVEL
- TOURIST INFORMATION
- HEALTH CARE
- MISCELLANEOUS






 Write Comment
Write Comment E-Mail
E-Mail Facebook
Facebook Twitter
Twitter  Print
Print 

