Dr Kajeĺs ayurvedic tablet trials to ’cure’ coronavirus to be suspended in Bengaluru
Dr Kajeĺs ayurvedic tablet trials to ’cure’ coronavirus to be suspended in Bengaluru
Mangalore Today News Network
Bengaluru, August 2, 2020: The ethics committee of the Bangalore Medical College and Research Institute (BMCRI) has decided to suspend the clinical trials for ayurvedic tablets ’Bhoumya and Saathmya’ which were introduced to "cure" coronavirus.
The medicines were introduced by a Bengaluru-based ayurvedic medical practitioner named Dr Giridhar Kaje, who allegedly went to the media claiming the success of the trials without presenting it to the ethics committee, media reports said.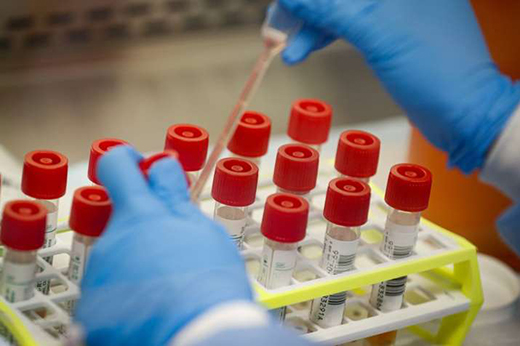
BMCRI, in a letter to Dr Kaje, said, "You are aware that the trial is still in its preliminary stage and no results have been placed before the ethics committee of BMCRI. Such being the factual position, you have gone to the public claiming success of the Trial. This is unacceptable and amounts to overstepping of one’s responsibility."
"You are called upon to immediately clarify in the media the factual position as otherwise we may be compelled to suspend the trial due to the unauthorised act of yours in misleading the public," the letter added.
The allegations have been denied by Dr Kaje, media reports said.
Last month, clinical trials for ayurvedic tablets were held between June 7 and 25 at the Victoria Hospital, which comes under BMCRI.
The trials were evaluating the efficacy and safety of both the tablets as an add-on therapy to the standard care given to Covid-positive patients, sources said.
- Case Against BJP Leader CT Ravi Over Social Media Post: Karnataka Poll Body
- Lok Sabha polls-24: 63% voter turnout recorded in phase 2
- LS elections 2024: Left rattled by allegations of CPI(M) veteran’s plans to join BJP
- Tejasvi Surya booked for ÔÇÖseeking votes in the name of religionÔÇÖ on polling day
- ÔÇśHe will shed tearsÔÇÖ: Rahul GandhiÔÇÖs sharp attack on PM Narendra Modi
- Lok Sabha elections: 4 men collapse and die amid Phase 2 voting in Kerala
- BJP becomes first Indian party to cross Rs 100 crore ad spend on Google
- 78 year old woman, on oxygen support, casts vote in Bengaluru
- Long queues outside Bengaluru hotel for free Benne Dosa, Ghee Laddu on polling day, Watch
- What happens if NOTA gets maximum votes? Supreme Court notice to poll body
- BJP candidate K Sudhakar booked for ÔÇÖBriberyÔÇÖ in Karnataka, Rs 4.8 Cr cash seized
- 2 Terrorists dead, 2 army personnel injured as gunbattle resumes in J&KÔÇÖs Baramulla
- Belthangady: Banjarumale records 100% voter turnout in Lok Sabha polls
- Dakshina Kannada, Udupi sees voter turnout of nearly 72%
- Mangaluru International Airport initiates work on precision approach lighting system
- Sandalwood star Rakshit Shetty casts his vote in Udupi, Watch
- Brisk polling across Dakshina Kannada constituency
- Ashta Mutt pontiffs cast vote in Udupi
- Udupi: Candidates Jayaprakash Hegde, Kota Srinivas Poojary exercise franchise
- Vittal: EVM technical glitch delays voting at Karopady booth
- Orange alert: Karnataka State Disaster Management cell gives animated advisory
- Lok Sabha elections 2024: Voting underway in 14 constituencies in Karnataka
- Mangaluru: Activists clash with media, police at Capitanio
- Karnataka Lok Sabha election: Voting timings, Key candidates and phase 2 polling constituencies
- Sullia: Rider dies in bike-jeep collision
- New residential complex for the judges inaugurated in Mangaluru
- Absconding accused nabbed after 8 years
- Truck with cylinders turns turtle in Beltangady
- Bhoota Kola artist dies of cardiac arrest
- Development of the country should be our goal: Ganesh Karnik
- Container truck gets stuck under Modankap railway bridge
- Truck crushes bike’s pillion rider near BC Road
- Head constable dies of heart attack
- Udupi: PDO dismissed over financial irregularities
- CREDAI to resume Skill Development Program for Construction Workers in Mangaluru
- John B Monteiro elected president of Rachana Catholic Chamber of Commerce & Industry
- Sudhanshu Rai elected district president of All College Student Association
- Chief Minister to visit Mangaluru, Udupi on August 1
- Nitte University awards PhD degree to Tina Sheetal D’Souza
- Sachitha Nandagopal honoured by CMTAI for Community Service
- CITY INFORMATION
- TRAVEL
- TOURIST INFORMATION
- HEALTH CARE
- MISCELLANEOUS




 Write Comment
Write Comment E-Mail
E-Mail Facebook
Facebook Twitter
Twitter  Print
Print 

